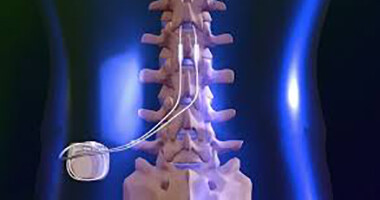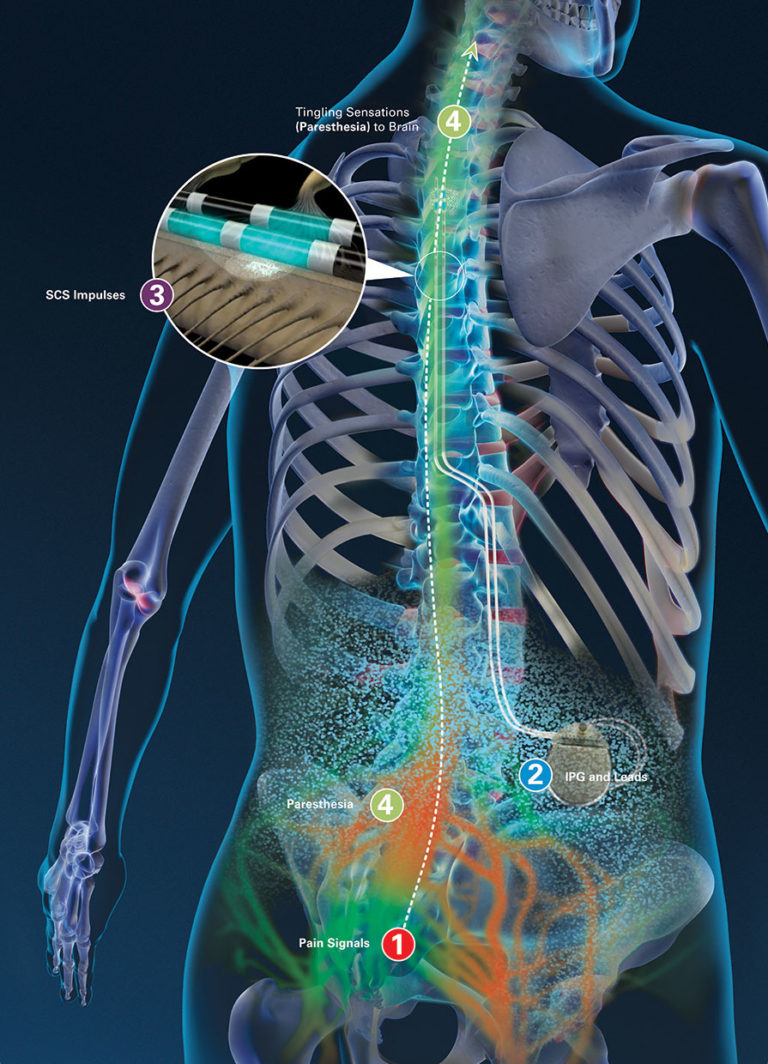
CSRWire - Abbott Strengthens Its Connected Care Technologies With Latest Neurosphere™ Mypath™ Digital Health App

Spinal cord stimulation for the treatment of peripheral neuropathic pain | Tidsskrift for Den norske legeforening

Abbott gains US FDA approval for Proclaim Plus spinal cord stimulation system - Spinal News International
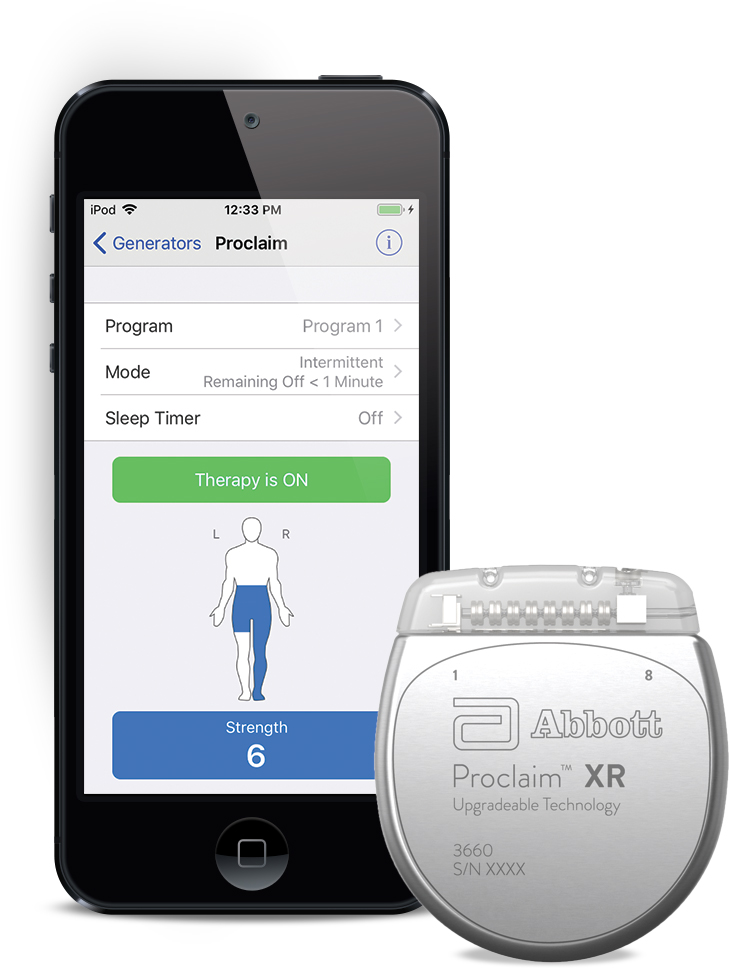
FDA Approves Abbott's "Low Dose," Recharge-Free Spinal Cord Stimulation System with up to Ten Year Battery Life* for People Living with Chronic Pain

Abbott on X: "We're giving doctors more options in tailoring treatment for chronic pain than ever before. Our Proclaim™ Plus spinal cord stimulation system, featuring FlexBurst360™ therapy, is now FDA approved. Learn